Green Hydrogen Technologies | Montel
Introduction
Hydrogen is an energy carrier, meaning that it can be easily and safely transported and stored from one place to another and then be processed/combusted in order to release energy once it has reached its desired location. A key thing that sets hydrogen as a method of energy production apart from others is that it not only produces no carbon emissions (once it has been isolated and obtained in its fuel form, unlike things like fossil fuels), but it produces no emissions whatsoever while still being a constant and reliable source of energy which can be easily adjusted to meet demand, unlike most forms of renewable/non-polluting energy currently in use (e.g. Solar and Wind).
However, the main issue with hydrogen as a form of energy production and transport is that the isolation of hydrogen to use as a fuel often has its issues; it sometimes takes more energy to separate and isolate hydrogen from the compounds that it is found in that it releases when burned, meaning the net energy production from isolating and burning hydrogen would be negative, and sometimes even emits certain types of greenhouse gas . If hydrogen is to become a stable and widely used source of energy, its future lies in finding a green and more efficient way to isolate it from its natural compounds.
Current methods for the isolation of hydrogen
Currently, hydrogen is usually isolated in one of two ways: steam-methane reforming (SMR) or electrolysis.
SMR is a process that isolates hydrogen through the reaction of hydrocarbons (in this case methane, CH₄) with water. An example of what this sort of reaction might look like can be seen below:
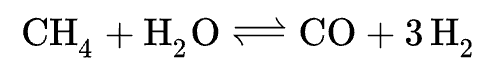
One thing you may immediately notice is that a byproduct of the reaction is Carbon Monoxide. Hydrogen produced from this type of isolation reaction is classed as ‘grey’ hydrogen as the process used to create it is not actually carbon neutral (this is true when working under the assumption that the carbon monoxide will turn into carbon dioxide in the atmosphere).
This process can also produce ‘blue’ hydrogen, which is the made with the exact same procedure except any carbon monoxide/carbon dioxide emissions which are released during its production are captured using capture and storage technology, meaning the process is closer to carbon-neutral but renders it more energy intensive; this can create practical issues as equipment can be expensive and the net energy obtained when the hydrogen is burned is lower, making the process less efficient and meaning it is not very widely used.
Another method that is often used to isolate hydrogen is the electrolysis of water. This process involves the release of hydrogen molecules at the cathode (negative electrode) following the attraction of a water molecule to said electrode. This type of hydrogen is classed as ‘green’ hydrogen (if the energy used to perform electrolysis is also green) and it is entirely carbon-neutral. However, electrolysis is a very expensive process due to the equipment needed and the scale on which it has to be performed in order to obtain any sizeable amount of hydrogen and it typically has an efficiency of 60-80% (meaning that 60-80% of the electrical energy input into the electrolysis is converted to chemical energy in the hydrogen). This unfortunately renders it unsuitable for large scale energy production and transport as there is an overall net energy loss, and it therefore would not actually create a net increase in electricity as what the energy it releases is less than what is needed to create it.
While there are other methods which can be used to isolate hydrogen aside from those mentioned above, most are held back by similar issues: cost, energy efficiency and carbon-emissions.
Alternative methods of production
In order to make hydrogen a viable long-term fuel source, new methods of isolation have to be developed on from where they currently stand; they either need to become more efficient or adjusted slightly to be suitable for large scale production.
One potential alternative is the isolation of hydrogen through biological processes, more specifically Biophotolysis, Photofermentation and/or Microbial Electrolysis.
Biophotolysis is a process carried out by various types of green algae and cyano bacteria that use light energy (i.e. the energy they can obtain from sunlight) to split water molecules into oxygen and hydrogen when in a sulfur deprived environment. However, the scalability of this process is yet to be proven and production on an industrial scale would be challenging due to the sensitivity of hydrogenase (the enzyme the organism above use to accelerate the division of water) to oxygen and its relatively low hydrogen yield when compared to methods of production currently being used.
Another biological method which could be used to scale up green hydrogen production is the use of exoelectrogenic bacteria (such as geobacter) which break down organic matter and create electrons as a by product. These electrons can then be used to reduce protons to form hydrogen atoms. This process is entirely green and does have a relatively high yield when compared to other biological process, but it is not currently suitable for large scale production operations as it requires external power inputs, meaning it is not entirely self-sufficient as a process and requires green energy to be produced elsewhere to fuel it. Additionally, this particular biological system is incredibly sensitive to temperature, PH, contaminants and oxygen levels. The combination of these factors ultimately means that this method is not available for industrial scale production; it could be adopted in the future when more efficient and developed technology is created, however, and these exoelectrogenic bacteria are in a good position to be a long term source of hydrogen for energy if the required technology appears on the market.
Outside of biological processes, hydrogen can also be isolated through the separation of methane, known as Methane Pyrolysis, which produces so called ‘blue’ hydrogen. This process involves heat/plasma as a reactant, rather than oxygen, and does not release any Carbon Dioxide into the atmosphere if done correctly with the proper precautions in place. The equation for this process can be seen below:
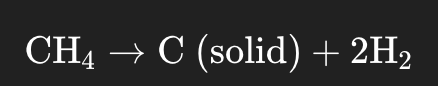
The splitting of methane, when done in optimum conditions, allows for any carbon which would usually be released in the form of a greenhouse gas to be captured and stored as a solid, meaning the process if entirely carbon neutral with regards to the atmosphere. This method of hydrogen production is currently limited and it still in its pilot stages but has the potential to become a mainstream isolation method as it is far more energy efficient than other methods and has a lower energy demand when compared to things like the electrolysis of water; however, there are a few reasons why this method is yet to become mainstream, the most significant of which is the lack of technological maturity and understanding in the field as the process is still in the very early stages of its commercialization, but in the future this process may well become a large scale operation.
Global Attitude
Attitudes towards hydrogen as an energy source vary across the world; however, in recent years, there has been a growth in support and understanding that hydrogen may be the long-term solution to growing global energy demands. For example, in 2020 the EU set out their own ‘hydrogen strategy’ which outlined that hydrogen could be an alternative to carbon-emitting fuels in sectors which are harder to de-carbonize with other methods of energy generation. The advantage that hydrogen has over many other non-carbon emitting energy sources is that is can produce a constant output of energy, making it more attractive to governments when compared to things like solar and wind energy as there is less risk of an outage or electricity shortage if hydrogen is implemented in a country compared to the other aforementioned methods of production.
Overall, what will drive the development and growth of hydrogen plants around the world, aside from an increased recognition and understanding that it is a potentially viable solution, in an increase in funding and research in the area, particularly in the form of government grants and projects, to allow new more efficient technologies to be developed and better understood so that they can one day be used on an industrial scale.
Fuel Cells and Transport
One of the many reasons that hydrogen is being favoured as a long term solution to growing global electricity demands is its relative safety and portability when compared to other mainstream types of electricity production.
Aside from the more obvious uses of hydrogen such as its combustion to release heat energy (with no carbon/greenhouse gas emissions), hydrogen can also be used in fuel cells which do not need to combust the hydrogen and instead convert it directly into electricity. They function by converting a hydrogen molecule into 2 protons (H+ ions) and 2 electrons, the latter of which are then made to flow through a circuit and generate an electrical current. The splitting of the hydrogen molecule is done by a catalyst, typically platinum, which first splits a hydrogen molecule into two hydrogen atoms by absorbing it and then oxidizes it by removing its electrons.
Hydrogen is also much easier to transport as an energy source compared to most other sources. Take nuclear fuel as an example, transporting nuclear fuel sources involves a large amount of planning, health and safety consideration and potential risks due to its dangerous and radioactive nature; hydrogen, on the other hand, can simply be transported in tanks (ensuring that there is nothing flammable around the hydrogen), and there is little to no danger if it is released into the atmosphere (when you compare the dangers associated with hydrogen in the atmosphere with the dangers associated with things like oil and uranium).
Conclusion
While in the long-term hydrogen appears to be an incredibly attractive energy source and storage method, it still has ways to go regarding the production/isolation of the hydrogen itself. Most methods for producing hydrogen as fuel are either inefficient (meaning it takes more energy to isolate the hydrogen than the hydrogen releases when it is burned/put in a fuel cell) or not entirely green, but as technology develops and the focus of research shifts more onto biological methods of production, I am sure that we will see hydrogen being used as a mainstream fuel source for electricity in coming years.
Bibliography
Consilium. (2020). Towards a hydrogen market for Europe: Council adopts conclusions. [online] Available at: https://www.consilium.europa.eu/en/press/press-releases/2020/12/11/towards-a-hydrogen-market-for-europe-council-adopts-conclusions/?utm_source=chatgpt.com [Accessed 26 Sep. 2025].
Consonni, S., Mastropasqua, L., Spinelli, M., Barckholtz, T.A. and Campanari, S. (2021). Low-carbon hydrogen via integration of steam methane reforming with molten carbonate fuel cells at low fuel utilization. Advances in Applied Energy, 2, p.100010. doi:https://doi.org/10.1016/j.adapen.2021.100010.
Energy.gov. (2024). Hydrogen Production: Natural Gas Reforming. [online] Available at: https://www.energy.gov/eere/fuelcells/hydrogen-production-natural-gas-reforming?utm_source=chatgpt.com.
Europa.eu. (2025). BASF: advancing CO2-free hydrogen production with electrolysis and methane pyrolysis | EU Chemicals Platform. [online] Available at: https://transition-pathways.europa.eu/chemicals/initiative/basf-advancing-co2-free-hydrogen-production-electrolysis-and-methane-pyrolysis [Accessed 26 Sep. 2025].
European Commission (2023). Hydrogen. [online] Energy. Available at: https://energy.ec.europa.eu/topics/eus-energy-system/hydrogen_en.
for, D. (2023). Hydrogen transport and storage networks pathway. [online] GOV.UK. Available at: https://www.gov.uk/government/publications/hydrogen-transport-and-storage-networks-pathway?utm_source=chatgpt.com [Accessed 26 Sep. 2025].
Georg, K. (2016). GER-BESITZKLAGEN DES ROMISCHEN. Wentworth Press.
Hygear. (2025). Tech: Steam Methane Reforming. [online] Available at: https://hygear.com/technology/steam-methane-reforming/?utm_source=chatgpt.com [Accessed 26 Sep. 2025].
Vardar-Schara, G., Maeda, T. and Wood, T.K. (2007). Metabolically engineered bacteria for producing hydrogen via fermentation. Microbial Biotechnology, 1(2), pp.107–125. doi:https://doi.org/10.1111/j.1751-7915.2007.00009.x.









