“Scientists have created a sponge-like technology using a Metal Organic Frameworks (MOFs) nanocomposite, which can quickly and inexpensively capture carbon dioxide from various sources, including the air itself.” | SciTechDaily
Last year, the Nobel Prize in Chemistry was awarded to 3 scientists, Richard Robson, Susumu Kitagawa, and Omar Yaghi, for their groundbreaking progress in the development of Metal-Organic Frameworks (MOFs). Initially discovered in the 1960s by Robson from a spark of creativity, it was Susumu Kitagawa and Omar Yaghi, who pioneered research into the material. This article will explain what MOFs are, why they are significant, and their potential for the future of environmental sustainability.
Chemical Explanation
MOFs are molecules made of metal ions linked with organic molecules. They are very large, 3D, crystalline structures, and their general structure is inspired by that of diamonds. The metal ions can be singular atoms or clusters of atoms, and depending on what metal is used, the properties of the MOF can change. Characteristics which can change include adsorption rate, chemical stability, thermal stability, hydrophobicity, hydrophilicity, and more. The versatility of MOFs allows them to be synthesised for various uses.
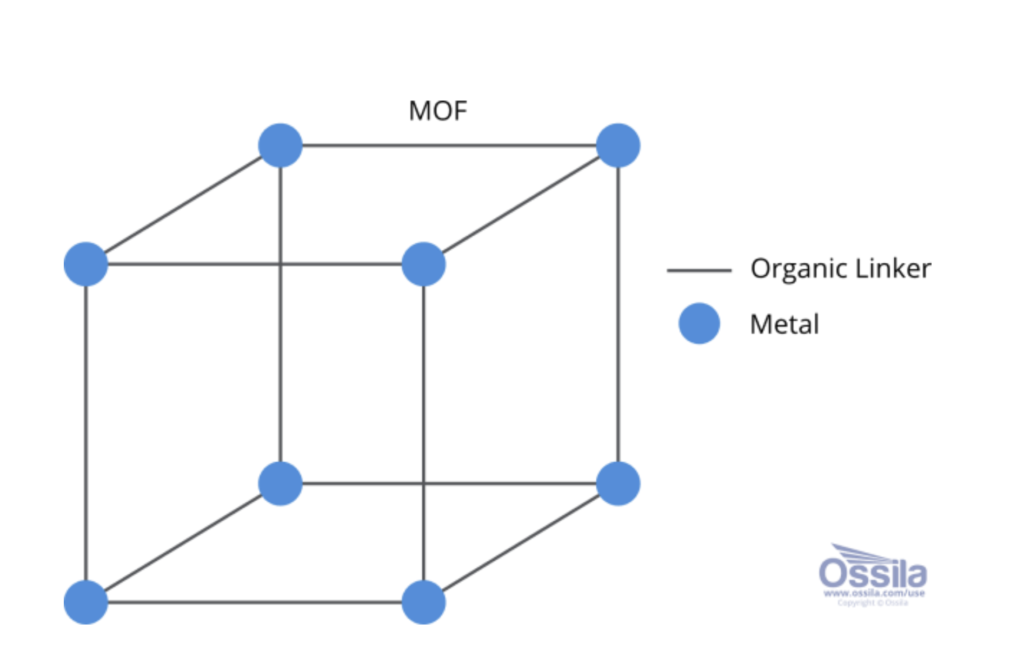
The ‘organic’ component of MOFs are the organic linkers or organic ligands. These are molecules or ions which form covalent bonds with a metal by donating a pair of electrons. This is what connects the structure of MOFs together.
Instead of a non-ionic molecule like water where an oxygen atom is bonded to two hydrogen atoms, and each bond has an electron from each atom, some covalent bonds can be formed by the two electrons coming from just one atom. This is known as a dative covalent bond, or a coordinate bond. (The dative case in Latin can translate as ‘to’ or ‘for’; an atom is donating a pair of electrons ‘to’ another atom.) This is different to ionic bonds because coordinate bonds are still sharing the pair of electrons.
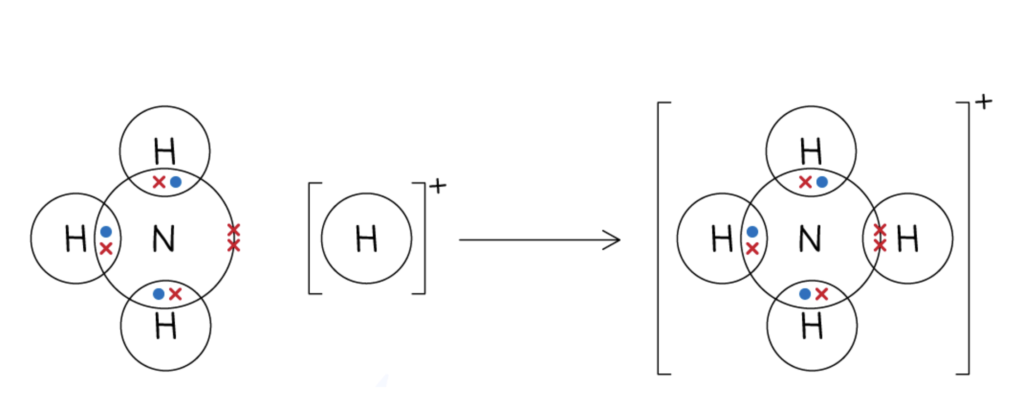
These organic ligands are often composed of oxygen or nitrogen atoms, because they are very good at donating electrons and therefore forming coordinate bonds to the metal atoms. This is because oxygen and nitrogen are two of the most electronegative atoms in the periodic table. Electronegativity is the measure of how much an atom attracts electrons to itself. This means that oxygen and nitrogen are the atoms which have the greatest tendency to attract electrons to fill their outer shell. Only fluorine tops these two in electronegativity, with a value of 4.0.
Commonly used organic linkers are carboxylic acids, and more specifically di/tricarboxylic acids, which have the functional group COOH. Di/tri-carboxylic acids have two or three of these functional groups on the molecule (more than one of these groups are needed or else you would not be able to link metal atoms on either side of the organic linker).
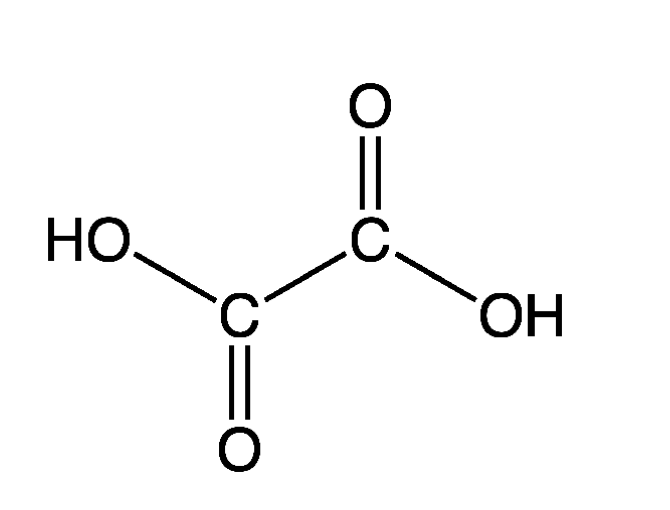
This is oxalic acid: the simplest dicarboxylic acid. It would have two electrons to spare, which will be used to form a coordinate bond with a metal ion. This is how the organic linkers hold the MOF together.
Environmental Application
MOFs are known to be extremely porous, and the length of the organic linker allows MOFs to have different pore sizes. These large cavities are what make MOFs so promising in the future of environmental science.
The first potential use is gas capture. Carbon capture is already an existing method of reducing carbon emissions. However, the current technology is expensive, energy demanding, and effective only to a limited extent. MOFs, on the other hand, are not specific to just carbon dioxide, but can be used to contain a wide range of gases just by changing the size of the pores and producing different species of MOFs. A chemical company, BASF, has already manufactured MOFs which ‘can capture CO2 emitted from various industrial sectors including hydrogen, cement, steel, aluminium and chemicals’. The most notable species of MOF which they manufacture is called ‘CALF20’, which is a zinc-based MOF, especially good at capturing industrial CO2. MOFs may prove to be more energy efficient, and more cost efficient; key factors to consider for sustainable gas capture.
Another potential use for MOFs is water purification. A growing challenge for humanity is providing clean water for all, and MOFs offer a solution. They can absorb particles and pollutants into their pores, and MOFs with suitable pore sizes can be specifically chosen and synthesised. Water is typically polluted by heavy metals from industrial processes which happen near water sources, such as mining, which releases toxic metals like mercury and lead. An example of an MOF which is used to purify industrially polluted water is from the UiO-66 branch of MOFs, which is zirconium based (atomic number 40). UiO-66 MOFs could allow water purification to become an easier, possibly cheaper, and more environmentally friendly process, which would be greatly beneficial to the environment and living standards.
However, bringing MOFs to industry is currently challenging due to the high costs of manufacturing, and ‘the lack of manufacturing methods that are scalable and environmentally sustainable.’ A detailed article by Fortune Business Insights states that reproducing and maintaining the stability of MOFs in industrial conditions is unreliable, which will compromise the efficiency and effectiveness of these materials. MOFs are relatively new and are emerging into the realm of chemical engineering and is still undergoing research.
There are predictions based on recent trends which ‘indicate a shift toward high stability, scalable, and multi-functional frameworks’ in the MOFs market. Over time, hopefully, the surge in technology such as AI is applied well to environmental science, to aid the development of technologies such as MOFs.
Overall, MOFs are an extremely promising class of materials for the environment, but also to various other applications from energy storage to drug delivery. Currently, cost of production, chemical stability, and infrastructure which is not suited to MOFs are key factors which hold back the potential for these materials. When more extensive research and funding are received, MOFs can have a great potential to be effective in addressing sustainability in a wide spectrum of sectors.
Works Cited:
“Metal Organic Frameworks Market Size, Share, Trends, 2034.” Consulting & Growth Advisory Services | Fortune
Business Insights, 27 Apr. 2026, www.fortunebusinessinsights.com/metal-organic-frameworks-market-111790.
“Metal-organic frameworks for environmental applications.” https://www.sciencedirect.com/ ,
www.sciencedirect.com/science/article/pii/S2666386421000333.
“MOF – Metal-organic Frameworks.” Chemical Catalysts and Adsorbents, chemical-catalysts-andadsorbents.basf.com/global/en/custom-catalysts/MOF.
“Nobel Prize in Chemistry 2025.” NobelPrize.org, www.nobelprize.org/prizes/chemistry/2025/press-release/
Ossila, www.ossila.com.
“What Is Stopping MOFs Being More Widely Used?” Structural Chemistry Data, Software, and Insights | CCDC,
www.ccdc.cam.ac.uk/discover/blog/what-is-stopping-mofs-being-more-widely-used/










